Current Issue
Beyond Pressure: Towards an Integrative Paradigm in Glaucoma Management
Honassys R. Rocha Silva*
Medical Director, Clínica do Olho Salvador, Bahia, Brazil
*Corresponding author: Honassys R. Rocha Silva, MD, Medical Director, Clínica do Olho Salvador, Bahia, Brazil, Phone: + 55 71 99258-0001, E-mail: [email protected]
Received Date: February 10, 2025
Published Date: March 16, 2026
Citation: Silva HRR, et al. (2026). Beyond Pressure: Towards an Integrative Paradigm in Glaucoma Management. Traditional Medicine. 7(1):29.
Copyright: Silva HRR, et al. © (2026).
Keywords: Glaucoma, Intraocular Pressure, Neuroprotection, Neuroinflammation, Mitochondrial Dysfunction, Integrative Medicine
INTRODUCTION
Glaucoma, a leading cause of irreversible blindness worldwide, is classically understood as a progressive optic neuropathy in which elevated intraocular pressure (IOP) is the primary modifiable risk factor [1]. This premise was consolidated by landmark studies, such as the Ocular Hypertension Treatment Study (OHTS), which unequivocally demonstrated that ocular hypotensive therapy delays or prevents the conversion of ocular hypertension to primary open-angle glaucoma (POAG) [2]. Consequently, global clinical practice has adopted IOP reduction as the cornerstone of treatment.
However, the persistent clinical observation of patients who continue to experience visual field damage progression, despite achieving established pressure targets, critically challenges this paradigm [3,4]. Such cases suggest that IOP, while undeniably crucial, represents only one facet of a much more complex pathophysiological cascade. The continued degeneration of retinal ganglion cells (RGCs) in scenarios of controlled IOP indicates the coexistence of other pressure-independent injury mechanisms. This realization compels us to re-evaluate the therapeutic approach, questioning whether the fixation on a single variable might be obscuring more comprehensive and effective therapeutic opportunities.
This realization is not entirely novel. To the best of our knowledge, Dada et al. were among the first to formally articulate the imperative of shifting the therapeutic focus beyond intraocular pressure, proposing that glaucoma management should target not only the eye, but the patient behind the eye, a concept encapsulated in their characterization of glaucoma as a condition of a “sick eye in a sick body”. Building upon this foundational insight, the present article proposes a broader and more systematic transition toward a patient-centered paradigm, defined operationally as an approach that integrates IOP control with the assessment and modulation of systemic, metabolic, genetic, and lifestyle factors that collectively determine neuronal vulnerability [5].
This article aims to outline the foundations of this integrative model, synthesizing evidence on non-pressure-related injury mechanisms and highlighting adjuvant interventions designed to strengthen retinal ganglion cell resilience and optimize systemic health.
This article advocates for expanding the focus of glaucoma management beyond intraocular pressure. It proposes a transition to a “patient-centered” paradigm, where pressure control is contextualized within a multifactorial, integrative approach oriented toward neuronal resilience and overall systemic health.
THE MULTIFACTORIAL PATHOGENESIS OF GLAUCOMA: BEYOND PRESSURE
The reductionist view of glaucoma as a purely pressure-driven disease is increasingly untenable. Growing evidence delineates a pathophysiological landscape where multiple cellular and systemic processes converge to promote neurodegeneration. The vulnerability of RGCs appears to be determined by a complex interplay between genetic susceptibility, mechanical stressors (IOP), and a host of other non-pressure-related factors.
Neuroinflammation and the Immune Response
Far from being an immune-privileged site, the eye exhibits a complex interaction with the immune system in both health and disease. In glaucoma, neuroinflammation has emerged as a central mechanism. Recent studies demonstrate that glaucoma is associated with a systemic pro-inflammatory profile, and not just a localized response. A 2024 serum proteomic analysis revealed that glaucoma patients have significantly lower levels of multiple neuroprotective proteins (such as TWEAK, CX3CL1, NT-3) and elevated levels of the inflammatory marker IL-22Rα1, suggesting that glaucoma may be the ocular manifestation of an underlying systemic disorder [6]. The chronic activation of microglia and astrocytes in the retina and optic nerve releases pro-inflammatory cytokines (e.g., TNF-α, IL-1β), perpetuating a cycle of neuronal damage [7]. The emerging connection with autoimmunity and the gut-retina axis deserves mention as a future research frontier that may further underscore the systemic nature of the disease [8].
Mitochondrial Dysfunction and Oxidative Stress
Retinal ganglion cells (RGCs) have an exceptionally high metabolic demand, making them critically dependent on mitochondrial function for ATP production and the maintenance of cellular homeostasis. Mitochondrial dysfunction is now recognized as an early and central event in the pathogenesis of glaucoma [9]. Genetic mutations associated with glaucoma (e.g., OPTN) can directly compromise mitophagy, the process of removing damaged mitochondria, leading to their accumulation and cell death [10]. The imbalance in mitochondrial dynamics (fusion and fission) and increased oxidative stress create a toxic environment that culminates in the apoptosis of RGCs. This convergence of defective protein folding, endoplasmic reticulum stress, and mitochondrial failure highlights promising therapeutic targets aimed at restoring cellular bioenergetics [9].
Excitotoxicity and Neurotransmitter Imbalance
Glutamate-mediated excitotoxicity, driven by the primary excitatory neurotransmitter in the retina, is another well-established mechanism of damage. Failure to reuptake glutamate from the synaptic cleft leads to hyperactivation of N-methyl-D-aspartate (NMDA) receptors on RGCs, resulting in an excessive influx of calcium and the activation of apoptotic pathways [11]. More recently, the role of other neurotransmitters, such as dopamine, has been elucidated. A 2023 study demonstrated that in experimental glaucoma, the expression of dopamine type 2 receptors (D2R) is increased in RGCs. The activation of these receptors exacerbates neuronal hyperexcitability and cell injury, while the activation of D1 receptors (D1R) confers neuroprotection [12].
DISCUSSION: TOWARDS AN INTEGRATIVE AND PATIENT-CENTERED APPROACH
The recognition of glaucoma’s multifactorial nature demands a paradigm shift: from a “pressure-target” approach to a “patient-centered” and integrative model. This new paradigm does not dismiss the importance of IOP control but rather contextualizes it within a broader strategy aimed at enhancing neuronal resilience and optimizing systemic health. Lifestyle interventions, previously considered marginal, gain prominence as evidence-based adjuvant therapies.
Nutrition and Neuroprotective Supplementation
Diet and nutritional supplementation offer promising avenues for modulating inflammation, oxidative stress, and cellular metabolism in glaucoma patients. Supplementation with B-complex vitamins (B6, B9, B12) and choline has been shown to confer robust neuroprotection in preclinical models of glaucoma, preventing neurodegeneration and preserving visual function.
Notably, this 2025 study clarified that elevated homocysteine, previously seen as a causal factor, is more likely a pathogenic consequence of dysfunctional one-carbon metabolism, and not a primary therapeutic target in itself [13].
Antioxidants, such as those found in diets rich in fruits and vegetables, showed in a 2025 meta-analysis the ability to reduce IOP and slow visual field progression [14]. Bioactive compounds such as resveratrol, curcumin, and Ginkgo biloba are also being investigated for their pleiotropic effects, which include neuroprotection and improvement of ocular blood flow [15].
Vitamin D has been extensively investigated in studies examining the relationship between circulating vitamin D levels and glaucoma. Evidence suggests that higher serum concentrations may exert a protective effect against the risk of developing the disease; however, no consistent association has been demonstrated between low vitamin D levels and glaucoma severity [16]. Similarly, Ascorbic acid (vitamin C) has been independently associated with a protective effect against primary open-angle glaucoma (POAG). A systematic review and meta-analysis by Ramdas et al. [17] demonstrated that, following correction for between-study heterogeneity, high dietary intake of vitamin C was associated with a statistically significant reduction of approximately 61% in the risk of developing POAG (OR 0.39; 95% CI: 0.23–0.67). The mechanistic basis for this association is thought to reside in the antioxidant properties of ascorbic acid within the trabecular meshwork, where oxidative stress is known to impair aqueous humor outflow facility and contribute to intraocular pressure elevation. Consistent with this hypothesis, dietary patterns characterized by high consumption of dark green leafy vegetables, a principal source of vitamin C, demonstrated a uniform protective association with glaucoma across all studies included in the meta-analysis [17].
In addition, the potential role of minerals in the management of patients with glaucoma should be highlighted, particularly magnesium. Magnesium L-threonate has been described as a modulator of neuroinflammation [18] and exhibits potential protective effects on retinal ganglion cells [19].
The implementation of a nutritional supplementation strategy must, however, be guided by safety considerations. Hypervitaminosis, particularly with fat-soluble vitamins such as A, D, E, and K, can lead to systemic toxicity when recommended doses are exceeded. Potential supplement-drug interactions also warrant careful evaluation: high-dose vitamin E may potentiate the effect of anticoagulants, increasing bleeding risk, while Ginkgo biloba can interfere with the metabolism of several drugs via the cytochrome P450 system. Accordingly, supplementation should be individualized, grounded in documented deficiencies or strong scientific rationale, and monitored by a healthcare professional to ensure patient safety and therapeutic efficacy.
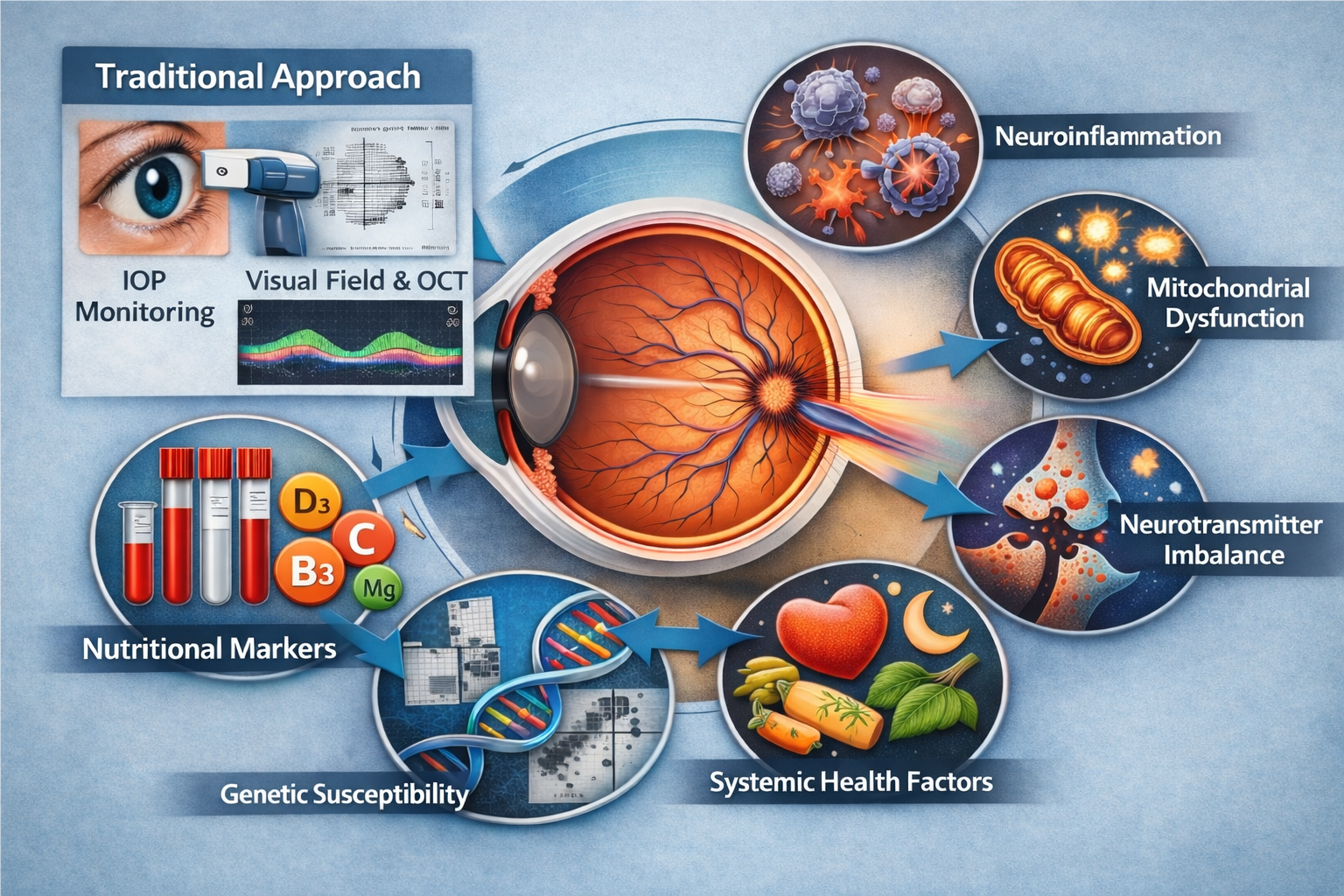
Although promising, the prescription of supplements must be judicious and grounded in evidence. Clinical guidance should focus on healthy dietary patterns and targeted supplementation where there is proven deficiency or a strong scientific rationale. Figure 1 illustrates the transition from a paradigm exclusively centered on intraocular pressure to an integrative model for the management of patients with glaucoma.
Figure 1: From traditional management to an integrative paradigm in glaucoma.
Schematic illustration comparing the traditional model of glaucoma management with an integrative, patient-centered approach. In the left panel (“Traditional Approach”), disease monitoring relies primarily on intraocular pressure (IOP) reduction and structural and functional assessment using optical coherence tomography (OCT) and automated visual field testing. In the integrative model, depicted around the optic nerve, glaucoma is conceptualized as a multifactorial optic neuropathy influenced by neuroinflammatory processes, mitochondrial dysfunction, neurotransmitter imbalance, systemic factors, and genetic susceptibility. In addition, nutritional and metabolic markers, including vitamin D3, vitamin B3, vitamin C, and magnesium, are considered potential modulators of neuronal vulnerability. This perspective supports the integration of pressure control with neuroprotective and systemic optimization strategies, aiming for a more comprehensive and personalized approach to glaucoma care. (Source: Generated by artificial intelligence. ChatGPT platform, version 5.2. February 10, 2026.)
Meditation and Stress Reduction
The mind-body connection in the context of glaucoma is gaining recognition. Chronic stress, with the consequent elevation of cortisol, is postulated as a factor that can contribute to the elevation of IOP and disease progression [20]. Interventions such as mindfulness meditation have shown remarkable efficacy as an adjuvant therapy. A 2024 meta-analysis concluded that meditation was the most effective relaxation technique, resulting in an average IOP reduction of approximately 32% [21]. A 2018 randomized clinical trial had already shown that meditation not only reduces IOP and stress biomarkers, but also positively modulates the expression of genes associated with glaucoma and improves quality of life [22]. These findings provide a solid evidence base for integrating stress reduction practices into the patient’s care plan.
In conclusion, glaucoma management is at a pivotal turning point. The exclusive reliance on IOP reduction, while essential, is an incomplete strategy that fails to address the complex neurodegenerative processes in a significant portion of patients. Accumulated evidence positions glaucoma not as a simple pressure-induced disease, but as a complex optic neuropathy where inflammation, metabolic dysfunction, and excitotoxicity play crucial roles.
The future of treatment lies in the clinician’s ability to adopt a holistic perspective, integrating molecular and systemic knowledge into daily clinical practice. The transition to a “patient-centered” paradigm implies evaluating and addressing individual risk factors, from genetic and metabolic profiles to lifestyle choices. Neuroprotective strategies aimed at strengthening RGC resilience and interventions that modulate systemic health, such as nutritional optimization and stress reduction, should be considered valuable adjuvant therapies, not marginal alternatives.
CONFLICT OF INTEREST
The author declares that the work was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
REFERENCES
- Schuster AK, Erb C, Hoffmann EM, Dietlein T, Pfeiffer N. The Diagnosis and Treatment of Glaucoma. Deutsches Ärzteblatt international. 27;117(13):225-234.
- Kass MA, Heuer DK, Higginbotham EJ, Johnson CA, Keltner JL, Miller JP, et al. (2002). The Ocular Hypertension Treatment Study: a randomized trial determines that topical ocular hypotensive medication delays or prevents the onset of primary open-angle glaucoma. Archives of ophthalmology.. 120(6):701-13.
- (2000). The Advanced Glaucoma Intervention Study (AGIS): 7. The relationship between control of intraocular pressure and visual field deterioration. The AGIS Investigators. 130(4):429-440.
- Leske MC, Heijl A, Hyman L, Bengtsson B, Komaroff E. (2004). Factors for Progression and Glaucoma Treatment: The Early Manifest Glaucoma Trial. Current Opinion in Ophthalmology. 15(2):102-106.
- Dada T, Ramesh P, Shakrawal J. (2020). Meditation: a polypill for comprehensive management of glaucoma patients. Journal of Glaucoma. 29(2):133-140.
- Okruszko MA, Szabłowski M, Zarzecki M, Michnowska-Kobylińska M, Lisowski Ł, Łapińska M, et al. (2024). Inflammation and Neurodegeneration in Glaucoma: Isolated Eye Disease or a Part of a Systemic Disorder?-Serum Proteomic Analysis. Journal of Inflammation Research. 17:1021-1037.
- Rolle T, Ponzetto A, Malinverni L. (2021). The Role of Neuroinflammation in Glaucoma: An Update on Molecular Mechanisms and New Therapeutic Options. Front Neurol. 11:612422.
- Yang Z, Tian D, Zhao X, Luo Y, Chen Y. (2024 ). The gut-retina axis: Uncovering the role of autoimmunity in glaucoma development. Heliyon. 10(15):e35516.
- Venkatesan A, Bernstein AM. (2025). Protein misfolding and mitochondrial dysfunction in glaucoma. Frontiers in Cell and Developmental Biology. 13:1595121.
- Jeong Y, Davis CO, Muscarella AM, Navarro HH, Deshpande V, Moore LG, et al. (2025). Glaucoma-associated Optineurin mutations increase transcellular degradation of mitochondria in a vertebrate optic nerve. 14:e103844.
- Feng L, Dai S, Zhang C, Zhang W, Zhu W, Wang C, et al. (2024). Ripa-56 protects retinal ganglion cells in glutamate-induced retinal excitotoxic model of glaucoma. Model of Glaucoma. Scientific Reports.14(1):3834.
- Yin N, Wang HN, Ding WW, Zhou H, Li SY, Miao Y, et al. (2023).Dopamine receptor-mediated roles on retinal ganglion cell hyperexcitability and injury in experimental glaucoma. Cellular Signalling. 109:110781.
- Tribble JR, Wong VHY, Stuart KV, Chidlow G, Nicol A, et al. (2025). Dysfunctional One-Carbon Metabolism Identifies Vitamins B6, B9, B12, and Choline as Neuroprotective in Glaucoma. Cell Reports Medicine. 6(5):102127.
- Bao J, Yu Y, Chen G, Hu C, Zhao C, Li X. (2025). Effect of antioxidants on primary open-angle glaucoma: a systematic review and meta-analysis. Frontiers in Pharmacology. 16:1625735.
- Zhu Y, Moksha L, Salowe R, Vrathasha V, Pham K, Aibo MI, et al. (2025). Integrating Neuroprotection, Antioxidative Effects, and Precision Medicine in Glaucoma Management with Bioactive Compounds. Biomedicine & Pharmacotherapy. 190:118319.
- Chan HN, Zhang XJ, Ling XT, Bui CH, Wang YM, Ip P, et al. (2022). Vitamin D and ocular diseases: a systematic review. International journal of molecular sciences, 23(8):4226.
- Ramdas WD, Schouten JSAG, Webers CAB. (2018). The Effect of Vitamins on Glaucoma: A Systematic Review and Meta-Analysis. Nutrients. 10(3):359.
- Fu C, Huang L, Lian C, Yue J, Lin P, Xu L, et al. (2025). Effects of long-term magnesium L-threonate supplementation on neuroinflammation, demyelination and blood–brain barrier integrity in mice with neuromyelitis optica spectrum disorder. Brain Research. 1846:149234.
- Elghobashy M, Lamont HC, Morelli-Batters A, Masood I, Hill LJ. (2022). Magnesium and Its Role in Primary Open Angle Glaucoma; A Novel Therapeutic?. Frontiers in Ophthalmology. 2:897128.
- Polski A, Mai A, Stagg B, Wirostko B. (2025). The Role of Stress Reduction and Mindfulness Meditation in Glaucoma Management: A Review. The Open Ophthalmology Journal. 19(1): e18743641340238
- Zaher O, Kuchtaruk AA, McGinnis ES, Paunic M, Malvankar-Mehta MS. (2024). Effect of Various Relaxation Techniques on the Intraocular Pressure of Patients with Glaucoma: Systematic Review and Meta-Analysis. Canadian Journal of Ophthalmology. 59(4):e343-e349.
- Dada T, Mittal D, Mohanty K, Faiq MA, Bhat MA, Yadav RK, et al. (2018). Mindfulness Meditation Reduces Intraocular Pressure, Lowers Stress Biomarkers and Modulates Gene Expression in Glaucoma: A Randomized Controlled Trial. Journal of Glaucoma. 27(12):1061-1067.
 Abstract
Abstract  PDF
PDF