Current Issue
Chemical Constituents of Ranunculus multifidus Aerial extract and Evaluation of Antimicrobial and Antioxidant Activities
Terfo Yilma Mikre1*, Teshome Fikre2 and Samuel Sileshi3
1Department of Medicinal Chemistry, School of Pharmacy, College of Health and Medical Sciences, Haramaya University, Harar, Ethiopia
2Applied Chemistry Department, School of Applied Natural Sciences, Haramaya University, Harar, Ethiopia
3Department of Pharmacology, School of Pharmacy, Bisidimo Hospital, Harar, Ethiopia
Corresponding author: Terfo Yilma Mikre, Department of Medicinal Chemistry, School of Pharmacy, College of Health and Medical Sciences, Haramaya University, Harar, Ethiopia, Phone: 0976760242, e-mail: [email protected]
Received Date: February 12, 2026
Publication Date: April 16, 2026
Citation: Mikre TY, et al. (2026). Chemical Constituents of Ranunculus multifidus Aerial extract and Evaluation of Antimicrobial and Antioxidant Activities. Traditional Medicine. 7(1):36.
Copyright: Mikre TY, et al. © (2026).
ABSTRACT
Background: Infectious and inflammation diseases are still a growing public health concern, especially with the emerging challenge of drug resistance to the current drugs. Ranunculus multifidus, commonly known as Gubduu caaffee (in Oromiffa, Ethiopia), was a perennial herb traditionally used to treat malaria, asthma, wound healing, etc. Objective: The present study aimed to extract the bioactive metabolites from aerial part of R. multifidus and investigate the biological activity and isolation of the essential oils from solvent fractions using analytical grade solvents. Methods: In view of its medicinal uses, the powdered aerial part was extracted with methanol and successively fractionated with n-hexane, chloroform, ethyl acetate, n-butanol, and aqueous using solvent-solvent portioning technique. The fractions were evaluated in vitro using the disc diffusion method against S. pyrogens, S. aureus, E. coli, and P. aeruginosa. The antifungal activities of extracts were tested using disc diffusion methods against two fungal strains. Radical scavenging power of extract and isolate was done using DPPH assay techniques. GC- MS analysis was used for isolation of solvent fraction essential oils. Results: The powdered aerial part was extracted with methanol and successively fractionated with n-hexane, chloroform, ethyl acetate, n-butanol, and aqueous using solvent-solvent portioning to afford 4.54, 10.87, 26.27, 15.15, and 38.08%, respectively. A phytochemical test revealed the presence of flavonoids, phenols, tannins, saponins, and steroids in both fractions of the plant. Strong inhibition observed against S. aureus (13.34 ±0.89 mm) and E. coli (11.55±1.1 mm) at 200 mg/ml of ethyl acetate fraction compared to ciprofloxacin as standard. Strong inhibition zone recorded for ethyl acetate fraction against C. albicans (14.5 ± 1.3 mm) and A. niger (16.93±1.11 mm) at 100 mg/ml compared to fluconazole (21 ±1.5 mm) and ketoconazole (23.43±0.65 mm) for strains respectively. The antiradical activities of each fraction were determined using the DPPH assay. The n-butanol and ethyl acetate fractions show (94.187 %) and (90.78%), respectively, and are comparable to ascorbic acid (98.59%) at 100 µg/mL. Using its spectacular microbial and radical scavenging power, the ethyl acetate fraction was selected for identification of compounds using GC-MS and afforded 45 compounds, including 9,12,15-Octadecatrienoic acid (Z, Z, Z) (24.09%), 9,12,15-Octadeca trienoic acid, methyl ester (23.46%), Methyl 9-cis,11-trans-octadecadienoate (7.07%), and hexadecanoic acid, methyl ester (12.69 %), as major compounds. Conclusion: The present finding of these pharmacologically important secondary metabolites from R. multifidus extracts brings the future attention of experts to the medicinal importance of the plant.
Keywords: Traditional Medicine, Ranunculus multifidus, Antimicrobial Activities, Antioxidant Effects, Phytochemical Screening
INTRODUCTION
The need for novel chemical compounds to treat human diseases is ever-increasing [1]. The rapid development of drug-resistant microbes, the discovery of new cases of life-threatening infections, and the constant recurrence of diseases have pushed for advances in the field of drug discovery [2]. Among the major strategies to alleviate the current situation is to search for new antibacterial, antifungal, and antioxidant agents from natural sources as an alternative to synthetic antibiotics. Plants are known to produce a variety of secondary metabolites to protect themselves against a variety of their own pathogens, and therefore, those with documented traditional use appear to be a rich source of new antibacterial and antioxidant drugs [3]. This prompts a revival in the search for antibacterial and antioxidant agents in natural products.
Medicinal plants have been used to treat a wide range of diseases throughout the history of human beings [4,5]. Today, it plays a role in primary health care as a therapeutic remedy in many developing countries. This is mainly because most of these herbals are available, affordable, and have little or no side effects [4,6]. In the 21st century, studies have focused on a new group of bioactive components in medicinal plants that have protective effects against cell oxidation and alleviate certain degenerative diseases [7].
Ranunculus multifidus is one of the species growing in Ethiopia and is locally called Gubduu caaffee in Afan Oromo and Etse siol in Amharic. It is a perennial herb that is being used in Ethiopia for the treatment of a wide array of diseases, including cancer [8], malaria [9], eczema, toothache, anti-inflammatory [10], stomach ache, wounds, eye diseases, urinary tract infection [11], tuberculosis (TB) [12], shingles, sores and sexually transmitted disease (STD) [13] and others. Some species in the genus Ranunculus were reported to contain secondary metabolites, including kaempferol-3-O-glucoside, tricin7-O-β-D-glucopyranoside, isoscopoletin, and Anemonin, β-sitosterol [14]. Protocatechuic acid, luteolin factors [15], ranuncoside [7], vanillic acid, and gallic acid [16]. Other phytocompounds also reported from this species at different times from different parts of the plants using different solvents and extraction techniques such as bilobetin, sternbin, methyl paraben, 4-O-D-glucopyranosyl-p-coumaric acid, linocaffein, kaya flavone, podocarpus flavone A, isoginkgetin, amentoflavone, robusta flavone-4′-methyl-ether, and others [15].
Despite the enormous traditional use of this species against various diseases, there is no information on the chemistry or antimicrobial and antioxidant studies of the aerial extracts of R. multifidus. Hence, in this study, we report the isolation of 45 compounds from the ethyl fraction of aerial extracts of R. multifidus. The antimicrobial and antioxidant activities of different aerial fractions are reported herein for the first time.
MATERIALS AND METHODS
Plant Material
The fresh aerial parts as well as botanical specimens of Ranunculus multifidus were collected and packed in the plastic bag from Sultana village, Bekoji Woreda, Oromia, Ethiopia, which is 224 km from Addis Ababa, in November 2021. The plant material was authenticated by Melaku Wondafrash, and a voucher specimen (Voucher No. TY001) was deposited in the National Herbarium, Department of Botany, Addis Ababa University, for future references.
Figure 1: The image aerial parts of Ranunculus multifidus forks (Picture taken by Terfo Yilma in Bekoji wereda, Oromia region)
Extraction
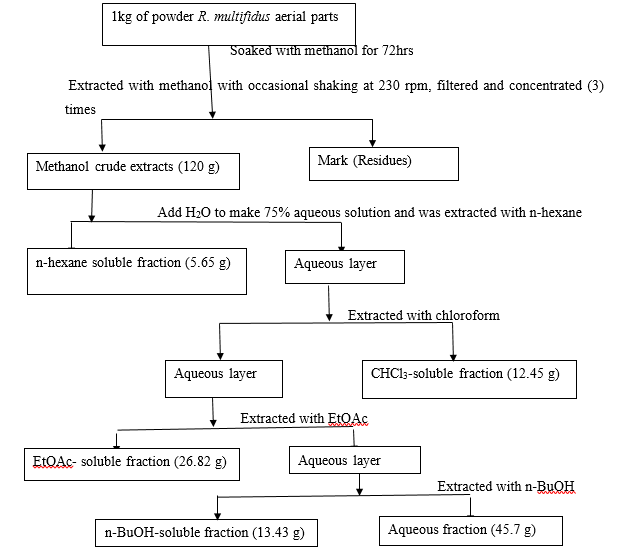
The air-dried aerial materials (1kg) of R. multifidus were milled into powder and extracted with 6 L of methanol for 72 hours with occasional shaking at room temperature using an orbital mini shaker at 230 rpm. The extracts were filtered and concentrated under reduced pressure using a rotary evaporator at 40 °C, leaving behind the methanolic extract (120g). Using liquid-liquid partitioning techniques, the methanolic extracts were fractionated using different solvents, starting from non-polar to polar solvents, according to Khan [17] with slight modifications. In the process, the methanolic extract was suspended in water and partitioned successively into n-hexane, chloroform, ethyl acetate, and n-butanol. The whole procedure of liquid-liquid fractionation is depicted in (Figure 2). Each fraction of solvents concentrated under reduced pressure using rotavapor and aqueous phase concentrated using lyophilizes to dry after putting overnight in a deep freezer at -20 °C to afford 4.54, 10.87, 26.27, 15.15, and 38.08%, respectively.
The whole procedure of liquid-liquid fractionation was shown in (Figure 2).
Figure 2: Schematic representation of liquid-liquid partitioning of aerial part of R. multifidus
Phytochemical Screening of Fractions
The word "Phyto" is the Greek word for plant. Phytochemicals are not only nutritive plant chemicals; they also have protective or preventive properties and also protect humans from host diseases [18]. Qualitative screening of various extracts of the plant was performed for the identification of various classes of chemical constituents in the plant based on the standard procedures with slight modifications as required [18–23].
Isolation of Compounds
Analytical TLC was applied to the detection and monitoring of compounds through a separation process and was used to optimize solvent systems for column chromatography. The band separation on the TLC describing the separation of compounds was detected under UV absorbance at 254 nm (absorption) and 365 nm (fluorescence), followed by spraying the TLC plates with vanillin-H2SO4 reagent and subsequent heating at 80 °C. The ethyl acetate extract (12 g) was adsorbed and fractionated over silica gel (200 g) by column chromatography with increasing polarity of EtOAc in n-hexane to afford 100 mL of each of the 52 major fractions. The first eight fractions were collected with 100 % n-hexane. Fr8-14, Fr15, Fr16-18, Fr19-21, Fr21, Fr22-27, Fr28-32, Fr33-36, F37-40, Fr41-44, Fr45-48, and Fr49-50 were eluted with 9:1, 4:1, 7:3, 3:2, 1:1, 2:3, 3:7, 1:4, 1:9, and 100% n-hexane: EtOAc, respectively, and the last 9:1 and 4:1 fraction were eluted using ethyl acetate to methanol. Fractions 15 (785 mg) after silica gel Column chromatography with 4:1 n-hexane: EtOAc as an eluent furnished a bright green, oily, non-separable spot with (30 mg. Fractions 28 _32 (453 mg) showed three spots on TLC, which were combined and subjected to column chromatography on silica gel with 85:15 EtAcO: MeOH as an eluent is still inseparable and has a yellow colour and oily nature (36 mg). Both fractions were submitted to GC-MS analysis to furnish 45 compounds together with some major.
Antimicrobial Investigation
Previously different scholars reported as this plant have different bioactivities, especially antibacterial and anti-malarial effects. According to Dilebo et al. [10] the leaf extracts of the plant show strong anti-inflammatory effects compared to indomethacin as standard anti-inflammatory. In the same scenario the anemonin isolated from hydroalcoholic extracts from leaf extracts of this plant show anti-malaria effects [9]. Having this in mind the present work tried to extracts the aerial arts of this plant and investigated its antibacterial, antifungal and antioxidant effects of different solvent extracts using standard methods.
Antibacterial Activity Test
Antimicrobial effects of aerial fractions of R. multifidus were investigated in vitro using the Kirby Bauer Disc diffusion technique against test bacterial and fungal strains [24]. The agar-well diffusion method is used to determine whether a bacterium is resistant or susceptible to fractions and pure compounds. This method was selected due to its simplicity, capacity to analyze multiple samples simultaneously, and ability to work well with defined inhibitors [25]. The activity of fractions was tested against four American Type Culture Collection (ATCC) bacterial strains. In the process, two Gram-positive bacteria, Streptococcus pyrogens (ATCC 12696) and Staphylococcus aureus (ATCC 25923), and two Gram-negative bacteria, Escherichia coli (ATCC 25922) and Pseudomonas aeruginosa (ATCC 27853), were used to evaluate the antibacterial activities. The medium was prepared using Muller-Hinton agar (MHA). The autoclaved medium um was poured into sterile plates (35–40 mL/plate), and the plates were allowed to solidify under sterile conditions at room temperature. After solidification, the plates were seeded with overnight-grown cultures of approximately 1.5 X 108 CFU/mL of bacterial strain and 104 CFU/mL of fungal strain by swabbing evenly onto the surface of the medium with a sterile cotton swab. Ciprofloxacin was used as a positive control, while DMSO was used as a negative control. The four bacterial strains were tested with 50, 100, and 200 mg/mL fraction concentrations using the paper disc-diffusion method [26] with slight modifications. For each concentration, 6 mm-diameter Whatman filter paper discs were soaked with a 100 µL solution of the above concentration for each fraction, and then these saturated paper discs were inoculated at the center of each petri dish having a bacterial lawn with the help of a sterile Cork borer [14]. The plates were incubated at 37 °C for 48 hours, and the inhibition zone that appeared around the paper disc in each plate was determined by measuring the diameter of the inhibition using a caliper.
Antifungal Activity Test
The anti-fungal activity was conducted against two common human pathogenic fungal strains, Candida albicans (ATCC 26555) and Aspergillus niger (ATCC 10864), which were incubated on Potato dextrose agar (PDA) for three days at 30 °C [27,28]. The anti-fungal activity was carried out by the agar-well diffusion method. Different dilutions of aerial fractions (10, 50, and 100 mg/mL) in DMSO were tested, while fluconazole and ketoconazole (50 µL, 1 mg/mL in DMSO) were used as the standard drugs. The zone of inhibition was recorded in the same fashion as above for each bacterial strain using a caliper. The results were expressed as the mean ± standard deviation. The bacterial and fungal strains were collected from Harar Health Science Medical Laboratory and Haramaya University Plant Pathology Department, respectively, and the work was done in collaboration with the medical microbiology laboratory and biology department at Haramaya University.
Antioxidant Activity Test
Evaluation of the DPPH radical scavenging assay (RSA)
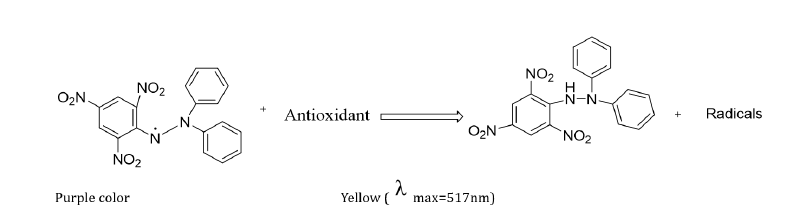
Antioxidants play an important role as health protection factors. Scientific evidence suggests that antioxidants reduce the risk of chronic diseases, including cancer and heart disease [3]. The DPPH assay was used to assess the free radical scavenging activity of fractions. With this method, the radical scavenging power of an antioxidant compound was measured with the principle of a decrease in absorbance due to the donation of hydrogen from the sample to the DPPH radical to produce stable DPPH-H, and the colour change from purple to yellow observed as shown in (Figure 3) below.
Figure 3: The structure of the DPPH radical and the scavenged radical
The radical-scavenging activities of the fractions were determined using the DPPH method [29]. In the process, the n-hexane, chloroform, ethyl acetate, n-butanol, and aqueous fractions of aerial extracts were dissolved in methanol to afford 1mg/mL. It was serially diluted in methanol to give a concentration of 500, 250, 125, and 62.5 μg/mL. To 1 mL of each concentration, 4 mL of DPPH (0.04% DPPH in MeOH) was added to make 100, 50, 25, and 12.5 μg/mL solutions. All samples were incubated in an oven at 37 °C for 30 min, and then absorbance was recorded at 517 nm using a UV-Vis spectrophotometer (Jenwey 6858, England). A reaction solution without DPPH was used as a blank, a DPPH solution as a control, and L-ascorbic acid as a standard. The percentage inhibition was calculated using the formula:
% Inhibition (RSA)= [A control –A test] / A control × 100…………………… (1)
RESULTS AND DISCUSSION
Phytochemical screening: Phytochemical screening tests were done to determine the classes of compounds present in each fraction following standard experimental protocols. The preliminary phytochemical screenings of different solvent fractions of aerial R. multifidus were tested. The results suggested that flavonoids, saponins, phenols, tannins, terpenoids, steroids, phytosterols, and glycosides are present within the fractions in different ranges (Table 1). The results obtained in this study suggest the identified phytochemicals may be the bioactive constituents of this plant, suggesting the plant is a valuable reservoir of bioactive compounds of substantial medicinal importance.
Table 1: Phytochemical screening of different fractions of aerial extracts of R. multifidus
|
Phytochemical screening |
Test type |
n-hexane fraction |
EtOAc fraction |
CHCl3 fraction |
n-BuOH fraction |
Aqueous Fraction |
|
Flavonoids |
Alkaline test |
- |
+ |
+ |
+ |
+ |
|
Saponins |
Foam test |
+ |
- |
+ |
- |
+ |
|
Phenols |
Alkaline test |
+ |
+ |
+ |
+ |
+ |
|
Tannins |
Ferric chloride test |
- |
+ |
+ |
+ |
- |
|
Terpenoids |
Salkowski test |
- |
+ |
- |
- |
+ |
|
Steroids |
- |
- |
+ |
- |
+ |
+ |
|
Phytostero |
Salkowski test |
- |
+ |
|
- |
+ |
|
Cardiac glycosides |
Keller-Killiani test |
- |
+ |
+ |
+ |
+ |
|
Alkaloids |
Wagner’s test |
- |
- |
+ |
+ |
- |
|
Anthocyanin |
- |
- |
+ |
- |
- |
+ |
|
Quinones |
- |
+ |
- |
+ |
- |
+ |
|
Reducing sugar |
Fehling’s test |
- |
+ |
+ |
+ |
+ |
|
Anthraquinone |
- |
- |
+ |
+ |
+ |
+ |
+ = the presence of phytochemical constituents - = the absence of phytochemical constituents
These secondary metabolites are known to be biologically active and play significant roles in the pharmacological effects of medicinal plants because the medicinal value of plants lies in these phytochemicals, which produce a definite pharmacological action on the human body. The presence of phenols could confer antioxidant and antimicrobial properties on the plant, as reported by various researchers from the genus.
Characterization of Isolated Compounds
The methanolic aerial extracts of R. multifidus were sequentially fractionated with n-hexane, CHCl3, EtOAc, and n-BuOH. The EtOAc extract was subjected to column chromatography for further purification following its better radical-scavenging activity and antimicrobial effects, as well as greater yields compared to other solvent fractions and afforded different compounds.
GC-MS Analysis
The essential oils obtained from ethyl acetate extracts of aerial parts of plant, after fractionation and esterification were analyzed by GC-MS at Adama Science and Technology University using an Agilent 6870 and an Agilent 5977 mass selective detector [MSD] operated in the EI mode (70 eV). Scan range 40_400 amu, scan rate 3.99 scans/s, and an Agilent Chem. Station data system. The GC column was HP-5 Fused silica capillary with a 5 % phenyl-polymethyl siloxane stationary phase film thickness of 0.25 µm a length of 30 m, and an internal diameter of 0.25 mM. The carrier gas was helium, with a column head pressure of 48.7 kPa and a flow rate of 1.0 mL/min. The inlet temperature was 20 °C, and the interface temperature was 280 °C. The GC oven temperature was used as follows: 60 °C initial temperature holds for 10 min. increased at 3 °C/min to 180 °C hold for 6 min. increased 5 °C/min to 220 °C A1 %w/v solution of the sample in hexane was prepared, and 1 µL was injected using a splitter of 1:20.
Identification of Compounds
The retention indices (RIs) for all of the essential oils were determined by co-injection of the sample with a mixture of the homologous series of C8–C25 n-alkanes. Identification of components was based on comparison of their mass spectra (MS) with those of the NIST MS Search 2.0 and Wiley 275 libraries and with those described by scholars [30].
Antibacterial activities of fractions from aerial extracts of R. multifidus
The antibacterial activity of the solvent fractions of the plant was determined by screening against two Gram-negative bacterial strains (E. coli and P. aeruginosa) and two Gram-positive strains (S. aureus and S. pyogenes) using the disc diffusion method. Ciprofloxacin is used as a positive control for selected strains. The zone of inhibition values indicated that all the fractions exhibited a varied range of antibacterial activities against all the tested bacterial strains. In comparison, the antibacterial activity of fractions at 50 mg/mL was weaker than at 100 mg/mL, which was less than 200 mg/mL, suggesting the activity displayed is dose-dependent in both aerial fractions (Figure 4). The result shows moderate concentrations relative to the fractions and standards used since ciprofloxacin has a broad spectrum of activities against pathogenic strains. The results of antibacterial activities against all strains for each compound were promising at all concentrations in concentration-dependent manner, as depicted in (Table 2).
Furthermore, the result showed better antibacterial activity against Gram-positive than the Gram-negative bacterial strains. The reason for the difference in sensitivity between Gram-positive and Gram-negative bacteria might be attributed to the differences in the morphological constitution of these microorganisms. Gram-negative bacteria have an outer lipopolysaccharide membrane, and this makes the cell wall impermeable to antibacterial chemical substances. Gram-positive bacteria, on the other hand, are more susceptible having only an outer peptidoglycan layer, which is not an effective permeability barrier. Therefore, the cell walls of Gram-negative bacteria are more complex in layout than those Gram-positive bacteria, acting as a diffusion barrier and making them less susceptible to antibacterial agents [31].
The antibacterial activity of compounds might be caused by the formation of reactive oxygen species (ROS), which leads to oxidative stress and subsequent cell death in pathogenic strains. The formation of ROS is a common antibacterial activity adopted by plant extracts Another possible mechanism for the antimicrobial activity of extracts and compounds is the attachment of extracts or compounds to the bacteria's cell membrane via electrostatic forces. This interaction may distort the plasma membrane structure and damage the bacterial cell integrity, resulting in the leakage of intracellular contents and cell death via apoptosis [32,33]. Further comparisons were also made with the literature reported for the antibacterial activities of this plant. The literature report showed that the methanol leaf extracts of R. multifidus and their chloroform and petroleum ether fractions show strong inhibition against different pathogens such as S. aureus DSM7346 (22.00±0.50 mm), P. aeruginosa DSM1117 (23.33±0.67 mm), E. coli ATCC25722 (21.67± 0.83 mm), and S. thyphimurium ATCC 13311 (22.67±0.83 mm), S. aureus (23.33±0.33 mm), P. aeruginosa (23.00±0.50 mm), E. coli (23.00±0.50 mm) and S. thyphimurium (23.67± 0.33 mm) and S. aureus (26.67±0.83 mm), P. aeruginosa (22.67±0.33 mm), E. coli (23.00±0.50 mm), S. thyphimurium (23.33±0.33 mm) at 200 mg/mL concentration with the solvent listed respectively [26]. The was comparable to the current result, even though the previous result was somehow greater than the current result due to the morphological difference of the strain used.
Table 2: Antibacterial activity of R. multifidus aerial fraction on selected bacterial strains
|
Fractions conc.(mg/ml) or µg/mL for pure compds.
|
Zone of inhibition in millimeter (mm) |
|||||
|
S. aureus |
E. coli |
P. aeruginosa |
S. pyrogens |
|||
|
Mean ±SD |
Mean ±SD |
Mean ±SD |
Mean ±SD |
|||
|
n-hexane fraction |
200 |
- |
- |
- |
- |
|
|
100 |
- |
- |
- |
- |
||
|
50 |
- |
- |
- |
- |
||
|
EtOAc fraction |
200 |
13.34±0.89 |
11.55±1.1 |
7.65±0.43 |
3.85±1.15 |
|
|
100 |
8.76±0.5 |
8.53±0.48 |
5.72±0.11 |
2.73±0.81 |
||
|
50 |
6.36±1.2 |
5.34±0.51 |
2.1±0.5 |
1.44±0.54 |
||
|
CHCl3 fraction |
200 |
7.13±0.56 |
6.86±0.28 |
- |
- |
|
|
100 |
|
4.09±0.13 |
- |
- |
||
|
50 |
- |
2.76±0.25 |
- |
- |
||
|
n-BuOH fraction |
200 |
7.13±0.56 |
6.43±0.61 |
- |
4.3±0.16 |
|
|
100 |
6.64±0.11 |
4.87±0.41 |
- |
2.87±0.81 |
||
|
50 |
4.39±0.30 |
3.91±0.11 |
- |
1.91±0.18 |
||
|
Aqueous fraction |
200 |
9.69±1.23 |
|
- |
- |
|
|
100 |
6.62±0.57 |
3.75±0.25 |
- |
- |
||
|
50 |
|
- |
- |
- |
||
|
Standard drug |
CIP(5µg/mL) |
22.28±0.83 |
24.32±0.61 |
23.52±0.48 |
18.81±0.34 |
|
Where, CIP stands for ciprofloxacin as positive control and the test was done in three replication.
Figure 4: Zone of inhibition of ethyl acetate extracts of Ranunculus multifidus on S.aureous and E.coli
Antifungal activities of aerial fractions of R. multifidus
The antifungal activities of fractions were evaluated for their antifungal effects against two human pathogenic fungal strains, C. albicans and A. niger. The antifungal test was performed using the agar disc diffusion method in the same manner with the same standard protocol as previously described by researchers [14]. The results obtained in this study revealed that all the studied fractions presented antifungal activity. The antifungal activities in terms of zone of inhibition in mm were measured for each fraction against selected strains and compared to the standard as shown in (Table 3). The chloroform and ethyl acetate fractions from the aerial extract had good inhibitory activity against both fungal strains. The n-hexane extract did not have any activity since all the inhibition zones had a diameter of 6 mm, which was the same as the diameter of the disc. The antifungal activities of fractions mostly target specific components of the fungal plasma membrane or target sterols in the synthesis machinery of cell wall components of fungal cells to combat fungal infections. The synergy of phytochemicals in the ethyl acetate fraction was also projected on its effect against selected fungal strains.
Table 3: Antifungal activity of R. multifidus aerial fraction and on selected fungal strains
|
Fraction conc. (mg/mL) and compounds in (µg/mL)
|
Zone of inhibition in millimeter(mm) |
||
|
C. Albicans |
A. Niger |
||
|
Mean ±SD |
Mean ±SD |
||
|
n-hexane fraction |
100 |
- |
- |
|
50 |
- |
- |
|
|
10 |
- |
- |
|
|
EtOAc fraction |
100 |
14.5 ± 1.3 |
16.93±1.11 |
|
50 |
7.38 ± 0.54 |
12.29±0.82 |
|
|
10 |
4.46±0.51 |
6.32±0.71 |
|
|
CHCl3 fraction |
100 |
5.74±0.63 |
- |
|
50 |
2.42±0.40 |
- |
|
|
10 |
- |
- |
|
|
n-BuOH fraction |
100 |
8.55 ±0.51 |
- |
|
50 |
4.89±0.10 |
- |
|
|
10 |
- |
- |
|
|
Aqueous fraction |
100 |
10.47±0.50 |
13.19±1.06 |
|
50 |
6.02±0.26 |
8.59±0.55 |
|
|
10 |
- |
4.09±0.11 |
|
|
Standard drug |
Flu(1mg/mL) |
21 ±1.5 |
|
|
Keto(1mg/mL) |
|
23.43±0.65 |
|
In all case the experiment done in triplicates. Flu=fluconazole, Keto= ketoconazole
Even though there was a lack of literature previously reported from the plant on antifungal activities, further comparison was also made with the literature reported for the compounds isolated from the genus. The Anemonin isolated from the ethyl acetate fraction of R. muricatus was reported to have inhibitory effects against A. niger [14] . This compound was also isolated from the leaves of R. multifidus and reported to have antimalaria and anti-leishmaniasis activities [9]. In the same scenario, the aqueous extracts of aerial parts from R. arvensis from the genus showed a very optimal inhibition zone against C. albicans (21 mm) and A. niger (16 mm), as reported before, which supports the current result of this paper [34].
DPPH radical scavenging assay: The DPPH assay was used to assess the free radical scavenging activity of the fractions. The DPPH antioxidant assay is based on the ability of 1,1-diphenyl-2-picryl-hydrazyl (DPPH), a stable free radical, to decolorize in the presence of antioxidants. The DPPH radical contains an odd electron, which is responsible for the absorbance at 517 nm and for the visible deep purple colour. A decrease in the absorbance of the reaction mixture indicates significant free radical scavenging activity of the extracts or fractions under investigation. In vitro antioxidant assays of crude extracts of R. multifidus revealed the presence of antioxidant potential. The percentage of inhibition was observed in all the antioxidant models in a concentration-dependent manner up to the given concentration. The data were compared to those obtained with the reference compound L-ascorbic acid. The relationship between different fractions of methanol extracts from the root of R. multifidus and their scavenging activity for the DPPH radical was shown in (Table 4) and (Figure 4). All investigated fractions showed moderate to high radical scavenging activities compared with the absorbance of the control (0.9118).
Table 4: % Radical scavenging activity of the aerial fraction of methanolic extract of Ranunculus multifidus
|
Conc.(µg/ml) |
Absorbance of fractions |
DPPH % inhibition |
||||||||
|
n-hexane |
EtOAc
|
CHCl3
|
n-BuOH
|
Aqueous |
n-hexane
|
EtOAc
|
CHCl3
|
n-BuOH
|
Aqueous |
|
|
12.5 |
0.95±0.01 |
0.17±0.31 |
0.77±0.26 |
0.16±0.03 |
0.70±0.02 |
3.75 |
81.03 |
15.44 |
82.56
|
23.67
|
|
25 |
0.91±0.12 |
0.17±0.44 |
0.76±0.29 |
0.12±0.04 |
0.60±0.03 |
0.86
|
81.47
|
16.21
|
86.95
|
34.42
|
|
50 |
0.90±0.01 |
0.13±0.03 |
0.74±0.07 |
0.11±0.09 |
0.51±0.65 |
1.40
|
85.53
|
18.62
|
88.37
|
43.96
|
|
100 |
0.83±0.01 |
0.08±0.34 |
0.48±0.09 |
0.05±0.02 |
0.23±0.01 |
8.64
|
90.79
|
47.45
|
94.19
|
74.99
|
|
Control 0.9118 |
|
|||||||||
Where, EtOAc = Ethyl acetate, n-BuOH = n-Butanol, CHCl3 = Chloroform, and the results were expressed in mean ±standard deviation and the test was done in triplicates.
The scavenging activity of fractions could be attributed to their hydrogen-donating ability. To compare the results of inhibition of fractions, the n-butanol (94.187%) and ethyl acetate (90.787%) fractions show a promising and comparable radical scavenging power relative to the other fractions relative to ascorbic acid (98.59%) at maximum concentration (100 µg/mL). The aqueous fraction also exhibited moderate hydrogen-donating ability at 100 µg/mL with a percentage inhibition of 74.99%. One the other way the n-hexane fraction shows almost very insignificant inhibition ability which might infer the fraction lacks polyphenolic groups in its constituents to donate single hydrogen to snatch the radical and break chain reaction. The root fractions of this plant show promising radical scavenging power, which might be attributed to the synergistic effects of phytochemicals in crude extracts.
Essential oil composition of ethyl acetate fractions of aerial part of R. multifidus
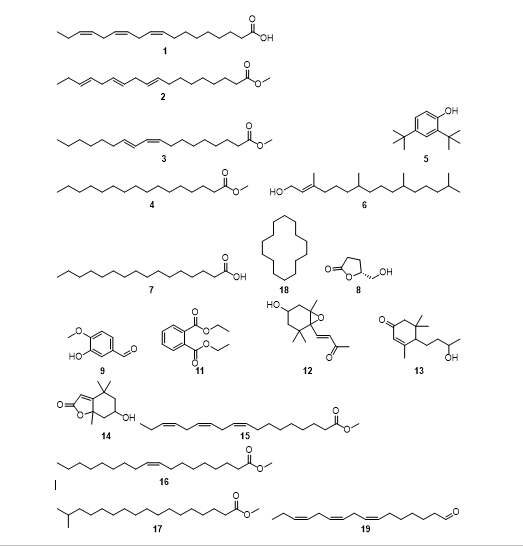
The ethyl acetate fractions of aerial extracts of R. multifidus were fractionated using column chromatography after TLC analysis of crude extracts to furnish colorless aromatic oil in a 0.89% yield. The essential oil was further analyzed with GC-MS to furnish 45 constituents (Table 5) and (Figure 5), accounting for about 99.9% of the essential oil compositions. Essential oil of the root part of R. multifidus were rich in 9,12,15-Octadecatrienoic acid, (Z,Z,Z)-(24.09%) (1), 9,12,15-Octadecatrienoic acid, methyl ester (23.46%) (2), Methyl 9-cis,11-trans octadecadienoate (7.07%) (3), Hexadecanoic acid, methyl ester (12.69 %) (4), Phenol, 2,4-bis(1,1-dimethylethyl)-(5.12 % ) (5), Phytol (4.34 %) (6), Palmitic acid P639 (4.29 %) (7), (5R)-5-(hydroxymethyl)-2-oxolanone (1.76 %) (8), Benzaldehyde, 3-hydroxy-4-methoxy- (0.51 %) (9), Cyclohexadecane P489 (0.66 %) (10), Diethyl phthalate (0.80 %) (11), 3-Buten-2-one, 4-(4-hydroxy-2,2,6-trimethyl-7-oxabicyclo[4.1.0]hept-1-yl)- (0.84 %) (12), 2-Cycloh exen-1-one, 4-(3-hydroxybutyl)-3,5,5-trimethyl-(0.77%) (13), 6-Hydroxy-4,4,7a-trimethyl-5,6,7,7a-tetrahydrobenzofuran -2(4H)-one (1.79%) (14), 9,12,15-Octadecatrienoic acid, methyl ester, (Z,Z,Z)-(0.73%) (15), 9-Octadecenoic acid (Z)-, methyl ester (0.65%) (16), Heptadecanoic acid, 16-methyl ester (1.37%) (17), Cyclotetradecane P391 (0.67%) (18), cis,cis,cis-7,10,13-Hexadecatrienal (0.56%) (19) are the major compounds which have area above 0.5%. Some compounds isolated from the aerial parts of R. multifidus using GC-MS root extracts were similar to the compounds isolated from [35].
There has also been reported Anemonin from the leaf extracts of R. multifidus, which has antimalarial effects [9]. The major compound, 9,12,15-Octadecatrienoic acid (Z,Z,Z) (ODA), is attributed to its cardiovascular-protective, anticancer, neuro-protective, anti-osteoporotic, anti-inflammatory, and anti-oxidative properties [36]. Recent pharmacological studies indicate that ODA has anti-metabolic syndrome, anti-cancer, anti-inflammatory, antioxidant, anti-obesity, neuro-protective, and is more specifically involved in the regulation of gut-microfloral functionalities. Studies, both experimental and clinical, indicate that ODA has antimetabolic syndrome effects. In short, ODA is potentially used to treat many diseases, but in-depth ADME studies are required to firmly re-establish its clinical efficacy and market potential [36]. Methyl 9-cis, 11-trans-octadecadienoate was also isolated from leaf extracts of Telfairia occidentalis and reported to have ethnomedicinal claims about the use of T. occidentalis in the management of sickle cell disorder [37]. Diethyl phthalate is widely used as a plasticizer and softener in pharmaceutical coatings, cosmetic additives, and insecticides. At high concentration ,there is a considerable decrease in the activity of GPX, superoxide dismutase, and GSH reductase [38]. This and other reports indicate that phytoconstituents from R. multifidus are promising for antimicrobial and antioxidant claims in rural parts of Ethiopia.
Figure 5: Chemical structure of compounds major compounds isolated from ethyl acetate aerial extracts of R. multifidus
Table 5: Essential oil constituents of ethyl acetate fractions of aerials part of R. multifidus
|
S. No |
Compounds name |
RI |
% Area |
|
1. |
3-Octanol |
1988 |
0.32 |
|
2. |
1-Nonanol |
2015 |
0.33 |
|
3. |
(3-amyloxiran-2-yl) methanol |
1981 |
- |
|
4. |
1(ax)-Formyl-1(eq)-(2'-propeny) cyclohexane |
2130 |
- |
|
5 |
(5R)-5-(hydroxymethyl)-2-oxolanone |
2104 |
1.76 |
|
6. |
1-Decanol |
2172 |
0.23 |
|
7. |
2,4-Decadienal, (E,E)- |
1953 |
- |
|
8. |
Benzaldehyde, 3-hydroxy-4-methoxy- |
2067 |
0.51 |
|
9 |
Phenol, 2,4-bis(1,1-dimethylethyl)- |
2059 |
5.12 |
|
10. |
(1R,2S,4R,6S)-4-Isopropenyl-1-methyl bicyclo [4.1.0] heptan-2-ol |
1944 |
- |
|
11 |
Cyclohexadecane P489 |
1300 |
0.66 |
|
12. |
Diethyl Phthalate |
2021 |
0.8 |
|
13. |
(10bR)-(+)-2-[(1S,2R,4R)-(2-Hydroxy-7,7-dimethylbicyclo[2.2.1]hepta-1-yl)methylsulfinyl]-8,9-dimethoxy-10b-methyl-5,6-dihydropyrrolo[2,1-a]isoquinolin-3-one |
1911 |
- |
|
14. |
2-Cyclohexen-1-one, 4-(3-hydroxy-1-butenyl)-3,5,5-trimethyl- |
1288 |
0.4 |
|
15 |
3-Buten-2-one, 4-(4-hydroxy-2,2,6-trimethyl-7-oxa bicyclo[4.1.0]hept-1-yl)- |
1687 |
0.84 |
|
16 |
2-Cyclohexen-1-one, 4-(3-hydroxybutyl)-3,5,5-trimethyl- |
2217 |
0.77 |
|
17. |
Acetic acid, bicyclo[2.2.1]hept-2-en-7-ylidene- |
1967 |
- |
|
18 |
6-Hydroxy-4,4,7a-trimethyl-5,6,7,7a-tetrahydrobenzofuran-2(4H)-one |
2105 |
1.79 |
|
19. |
(S,E)-4-Hydroxy-3,5,5-trimethyl-4-(3-oxobut-1-en-1-yl)cyclohex-2-enone |
2203 |
0.28 |
|
20. |
Cyclohexadecane P489 |
1742 |
1.25 |
|
21. |
(10bR)-(+)-2-[(1S,2R,4R)-(2-Hydroxy-7,7-dimethylbicyclo[2.2.1]heptan-1-yl)methylsulfinyl]-8,9-dimethoxy-10b-methyl-5,6-dihydropyrrolo[2,1-a]isoquinolin-3-one |
1878 |
- |
|
22. |
Neophytadiene |
2121 |
0.47 |
|
23. |
2-Pentadecanone, 6,10,14-trimethyl- |
2026 |
0.39 |
|
24. |
Phthalic acid, isobutyl tridec-2-yn-1-yl ester |
1823 |
0.21 |
|
25. |
2,4-Di-tert-butylphenol mesylate |
1976 |
- |
|
26. |
Neophytadiene |
1290 |
- |
|
27 |
9,12,15-Octadecatrienoic acid, methyl ester, (Z,Z,Z)- |
2155 |
0.73 |
|
28. |
9-Hexadecenoic acid, methyl ester, (Z)- |
2194 |
0.49 |
|
29 |
Hexadecanoic acid, methyl ester |
1948 |
12.69 |
|
30. |
cis,cis,cis-7,10,13-Hexadecatrienal |
2204 |
0.56 |
|
31. |
Palmitic acid P639 |
1894 |
4.29 |
|
32. |
Cyclohexadecane P489 |
1973 |
1.02 |
|
33. |
1-Ethynylcyclopentanol |
2040 |
- |
|
34. |
Heptadecanoic acid, methyl ester |
2150 |
0.21 |
|
35. |
(E)-2-nonadecene |
1964 |
0.22 |
|
36 |
Methyl 9-cis,11-trans-octadecadienoate |
1676 |
7.07 |
|
37 |
9,12,15-Octadecatrienoic acid, methyl ester |
1828 |
23.46 |
|
38. |
9-Octadecenoic acid (Z)-, methyl ester |
1846 |
0.65 |
|
39. |
Phytol |
1963 |
4.35 |
|
40. |
Heptadecanoic acid, 16-methyl-, methyl ester |
2080 |
1.37 |
|
41 |
9,12,15-Octadecatrienoic acid, (Z, Z, Z)- |
2238 |
24.09 |
|
42. |
Linoleic acid @P785 |
2093 |
0.31 |
|
43. |
Cyclotetradecane P391 |
1966 |
0.67 |
|
44. |
Octadecane P630 |
1531 |
- |
|
45. |
Linoleic acid @P785 |
1713 |
- |
RI: retention index. Results expressed as % area. Those compounds with % area beyond 0.2 % were excluded in this report
CONCLUSION
After esterification and GC-MS analysis, 45 chemicals were isolated from an ethyl acetate extract of R. multifidus using silica gel column chromatography. The majority of the main com pounds were discovered in the genus Ranunculus for the first time. The findings provided herein also indicated that the EtOAc extract of R. multifidus aerial exhibited strong antibacterial and antifungal activity when compared to ciprofloxacin and fluconazole, which were utilized as positive controls. The antioxidant activities of EtOAc extract were substantial when compared to that of ascorbic acid, showing the potential of this species aerial extracts as a natural antioxidant candidate. As a result, the biological activities demonstrated by the EtOAc extract and isolated chemicals from the aerial of R. multifidus support the traditional usage of this plant against various bacterial illnesses. Additional comprehensive studies, including in vivo antibacterial and antifungal activity testing, are required before making a final determination on the plant's potential candidature for formulation and pharmaceutical purposes.
STATEMENTS
Acknowledgments
The authors are thankful for the facilities provided by the University of Haramaya for extraction and biological study. Adama Science and Technology University for giving me a chance at postgraduate study and GC-MS analysis of a sample. This work was funded by Adama Science and Technology University under Grant (ASTU/AS-R/003/2020) and the World Academy of Sciences (TWAS) and the United Nations Educational, Scientific, and Cultural Organization (UNESCO) under the TWAS Research Grant (RGA No. 20-274 RG/CHE/AF/AC_G-FR3240314163).
Data Availability
The GC-MS data used for the interpretation of compounds in this study are included as supplementary information files. Other data used to support the findings of this study are available from the corresponding author upon request.
Conflicts of Interest
The authors declare that they have no conflicts of interest regarding this work or the publication of this paper.
Author contributions
All authors contributed equally in this manuscript from idea generations to manuscript submissions.
REFERENCES
- Zeleke D, Eswaramoorthy R, Belay Z, Melaku Y. (2020). Synthesis and antibacterial, antioxidant, and molecular docking analysis of some novel quinoline derivatives. Journal of Chemistry. 1:1324096.
- Muluye AB, Desta AG, Abate SK, Dano GT. (2019). Anti-malarial activity of the root extract of Euphorbia abyssinica (Euphorbiaceae) against Plasmodium berghei infection in mice. Malaria Journal. 18(1):1-8.
- Kalbessa A, Dekebo A, Tesso H, Abdo T, Abdissa N, Melaku Y. (2019). Chemical constituents of root barks of Gnidia involucrata and evaluation for antibacterial and antioxidant activities. Journal of Tropical Medicine. 2019:8486214.
- Anza M, Endale M, Cardona L, Cortes D, Eswaramoorthy R, Zueco J, et al. (2021). Antimicrobial activity, in silico molecular docking, admet and dft analysis of secondary metabolites from roots of three ethiopian medicinal plants. Advances and Applications in Bioinformatics and Chemistry. 14:117-132.
- Galma W, Endale M, Getaneh E, Eswaramoorthy R, Assefa T. (2021). Antibacterial and antioxidant activities of extracts and isolated compounds from the roots extract of Cucumis prophetarum and in silico study on DNA gyrase and human peroxiredoxin 5. BMC Chem. 15(1):32.
- Taher RF, Karmalawy AA Al, Abd AI, Maksoud E, Khalil H, Hassan A, Melaku Y. Two new flavonoids and anticancer activity of Hymenosporum flavum: in vitro and molecular docking studies. 2021;10(4):443-458.
- Raziq N, Saeed M, Ali MS, Zafar S, Shahid M, Lateef M. (2017). A new glycosidic antioxidant from Ranunculus muricatus L.(Ranunculaceae) exhibited lipoxygenasae and xanthine oxidase inhibition properties. Natural Product Research. 3;31(11):1251-1257.
- Ayele TT. (2018). A Review on Traditionally Used Medicinal Plants / Herbs for Cancer Therapy in Ethiopia : Current Status , Challenge and Future Perspectives.. Organic Chemistry: Current Research. 7(2):192.
- Sirak B, Mann L, Richter A, Asres K, Imming P. (2021). In Vivo Antimalarial Activity of Leaf Extracts and a Major Compound Isolated from Ranunculus multifidus Forsk. Molecules. 26(20):6179.
- Dilebo J, Gebre-mariam T, Asres K. (2010). Anti-inflammatory Activities of Extracts of Some Traditionally Used Medicinal Plants in Ethiopia on Carrageenan-Induced Mouse Paw Oedema. Ethiopian Pharmaceutical Journal. 28(1):32-38.
- Hashemi N, Ommi D, Kheyri P, Khamesipour F, Setzer WN, Benchimol M. (2021). International Journal for Parasitology : Drugs and Drug Resistance A review study on the anti- trichomonas activities of medicinal plants. International Journal for Parasitology. Drugs and Drug Resistance.15:92-104.
- Madikizela B, Kambizi L, Mcgaw LJ. (2017). An ethnobotanical survey of plants used traditionally to treat tuberculosis in the eastern region of O.R . Tambo district, South Africa. South African Journal of Botany. 109:231-236.
- Wet H De, Nzama VN, Vuuren SF Van. (2012). Medicinal plants used for the treatment of sexually transmitted infections by lay people in northern Maputaland, KwaZulu-Natal Province, South Africa. South African Journal of Botany. 78:12–20.
- Nazir S, Li B, Tahir K, Khan A, Ul Z, Khan H. (2013). Antimicrobial activity of five constituents isolated from Ranunculus muricatus. Journal of Medicinal Plants Research. 7(47):3438-3443.
- Aslam MS, Choudhary BA, Uzair M, Ijaz AS. (2012). The genus Ranunculus: a phytochemical and ethnopharmacological review. International Journal of Pharmacy and Pharmaceutical Sciences. 4(suppl 5):15-22. 4(5):15-22.
- Deng KZ, Xiong Y, Zhou B, Guan YM, Luo YM. (2013). Chemical Constituents from the Roots of Ranunculus ternatus and their Inhibitory Effects on Mycobacterium tuberculosis. 11859–11865.
- Khan S, Nisar M, Rehman W, Khan R, Nasir F. (2010). Anti-inflammatory study on crude methanol extract and different fractions of Eremostachys laciniata. Pharmaceutical Biology. 48(10):1115–1118.
- Kumar BVM. (2022). Screening of Some Medicinal Plants for Phytochemicals and Antibacterial Activities. 12(4):143-147.
- Anandharaj B, Sathya M, Maheshwari M, Priyadharshini B, Arun VP, Ramkumar R. (2021). Preliminary Phytochemical Screening of Natural Anti-dandruff plant Phylanodiflora (L.) Greene (Lippianodiflora). International Journal of Research Publication and Reviews. 2(7):1930–1932.
- Dey S, Dey N, Ghosh AK. (2010). Phytochemical Investigation and Chromatographic Evaluation of the Different Extracts of Tuber of Amorphaphallus paeoniifolius (ARACEAE). International Journal of Research in Pharmaceutical and Biomedical Sciences. 1(5):150–157.
- Ayoola GA, Coker HAB, Adesegun SA, Adepoju-bello AA, Obaweya K, Ezennia EC, et al. (2008). Method Spots Test. Tropical Journal of Pharmaceutical Research.7:1019–1024.
- Chintalapani S, Swathi MS, Narasu ML. (2018). Phytochemical screening and in vitro antioxidant activity of whole plant extracts of sesuvium portulacastrum l. Asian Journal of Pharmaceutical and Clinical Research. 11(1):322–327.
- Geetha TS, Geetha N. (2014). Phytochemical screening, quantitative analysis of primary and secondary metabolites of Cymbopogan citratus (DC) stapf. Leaves from Kodaikanal hills, Tamilnadu. International Journal of PharmTech Research. 6(2):521–529.
- Khalid S, Majid SA, Akram MA. (2019). The prophylactic effect of Ranunculus laetus (Wall)-mediated silver nanoparticles against some Gram-positive and Gram-negative bacteria. Bulletin of the National Research Centre. 43(1):18.
- Manoharan A, Pai R, Shankar V, Thomas K, Lalitha MK. (2003). Comparison of disc diffusion & E test methods with agar dilution for antimicrobial susceptibility testing of Haemophilus influenzae. The Indian Journal of Medical Research. 117:81-87.
- Begashawu T, Tariku Y, Bacha K. (2016). Antibacterial activity of selected medicinal plants used in South-western Ethiopia. African Journal of Microbiology Research. 10(46):1961–1972.
- Tran HNH, Graham L, Adukwu EC. (2020). In vitro antifungal activity of Cinnamomum zeylanicum bark and leaf essential oils against Candida albicans and Candida auris. Appl Microbiol Biotechnol. 104(20):8911–8924.
- Gizaw A, Marami LM, Teshome I, Sarba EJ, Admasu P, Babele DA, et al. (2022). Phytochemical screening and in vitro antifungal activity of selected medicinal plants against Candida albicans and Aspergillus niger in West Shewa Zone, Ethiopia. Advances in Pharmacological and Pharmaceutical Sciences. 2022:3299146.
- Rajesh P, Natvar P. (2011). In vitro antioxidant activity of coumarin compounds by DPPH, Super oxide and nitric oxide free radical scavenging methods. Journal of Advanced Pharmacy Education and Research. 1:52–168.
- Moniruzzaman, Shahinuzzaman, Haque A, Khatun R, Yaakob Z. (2015). Gas chromatography mass spectrometry analysis and in vitro antibacterial activity of essential oil from Trigonella foenum-graecum. Asian Pacific Journal of Tropical Biomedicine 5(12):1033-1036.
- Carvalho RS, Carollo CA, de Magalhães JC, Palumbo JMC, Boaretto AG, Nunes e Sá IC, et al. (2018). Antibacterial and antifungal activities of phenolic compound-enriched ethyl acetate fraction from Cochlospermum regium (mart. Et. Schr.) Pilger roots: Mechanisms of action and synergism with tannin and gallic acid. South African Journal of Botany. 114:181–187.
- Demissie MG, Sabir FK, Edossa GD, Gonfa BA. (2020). Synthesis of zinc oxide nanoparticles using leaf extract of lippia adoensis (koseret) and evaluation of its antibacterial activity. Journal of Chemistry. (1):7459042.
- Desalegn T, Murthy HCA, Adimasu Y. (2021). Medicinal Plant Syzygium Guineense (Willd) DC Leaf Extract Mediated Green. Ethiopian Journal of Science and Sustainable Development. 8(1):1-12.
- Hachelaf A, Zellagui A, Touil A, Rhouati S. (2013). Chemical composition and analysis antifungal properties of Ranunculus arvensis L. Pharmacophore. 4(3):89–91.
- Asmerom D, Kalay TH, Araya TY, Desta DM, Wondafrash DZ, Tafere GG. (2021). Medicinal Plants Used for the Treatment of Erectile Dysfunction in Ethiopia : A Systematic Review. 6656406.
- Parvathi K, Kandeepan C, Sabitha M, Senthilkumar N, Ramya S, Boopathi N, et al. (2022). In-silico Absorption, Distribution, Metabolism, Elimination and Toxicity profile of 9,12,15-Octadecatrienoic acid (ODA) from Moringa oleifera. Journal of Drug Delivery and Therapeutics. 12(2-S):142-150.
- Cyril-Olutayo MC, Agbedahunsi JM, Akinola NO. (2019). Studies on the effect of a nutritious vegetable, Telfairia occidentalis, on HbSS blood. Journal of Traditional and Complementary Medicine. 9(2):156-162.
- Premjanu N, Jaynthy C. (2014). Antimicrobial activity of diethyl phthalate: An insilico approach. Asian Journal of Pharmaceutical and Clinical Research 7(4):141-142.
 Abstract
Abstract  PDF
PDF